Published on 30.07.2025
Autoimmune lymphoproliferative syndrome (ALPS) is a rare, non-infectious, non-malignant genetic disease in which certain T lymphocytes, known as "double-negatives", accumulate abnormally in the blood and lymphoid organs (spleen and lymph nodes). Autoimmune manifestations and a high risk of developing lymphoma have also been observed in the majority of ALPS patients. Within our genome, each gene is present in 2 copies, each coming from one of the biological parents.
The Immunogenetics of Paediatric Autoimmune Diseases laboratory, headed by Frédéric Rieux-Laucat at Institut Imagine, described the first mutations of the FAS gene in ALPS 30 years ago. At the time, these were mutations inherited from a parent affecting only one copy of the FAS gene: these are known as heterozygous or monoallelic mutations. This team then showed that FAS mutations can appear spontaneously in lymphocyte progenitors during foetal development: these new mutations are known as somatic mutations.
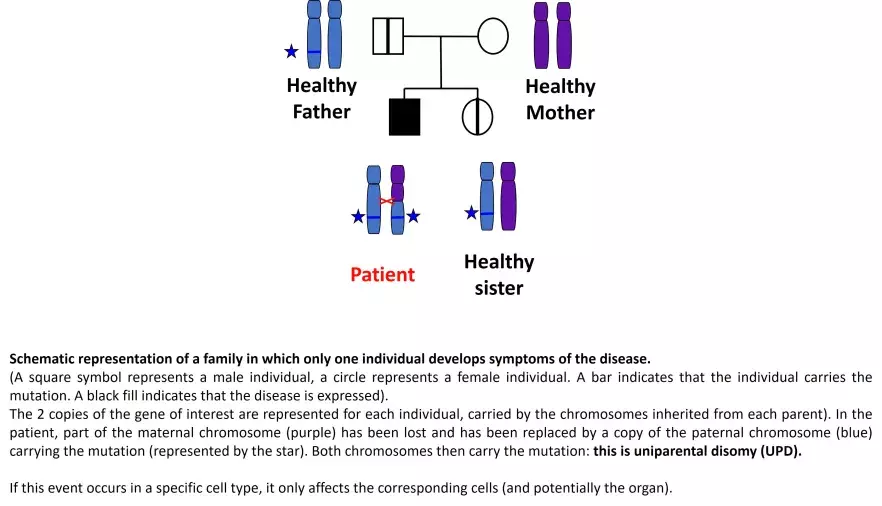
It also happens that the mutations causing ALPS are inherited without the parent who passed on the mutation showing any symptoms, as if the expression of the disease could 'skip a generation'.
In more recent work, the team showed that, in these families, the sick individuals carry, in addition to the inherited mutation, a 'somatic event' on the second copy of the FAS gene, and only in the abnormal T lymphocytes. These somatic events correspond to a loss of the healthy copy of the FAS gene. The missing piece of chromosome is replaced by a copy of the equivalent piece of chromosome from the other parent, which duplicates the mutated copy of the gene: the mutation is then present in both copies of the gene (see link ref Magerus J Clin Invest 2011). This "somatic event" is called uniparental disomy (UPD).
The FAS gene encodes the protein of the same name, FAS, which plays a major role in apoptosis, a form of programmed cell death that, in particular, makes it possible to eliminate undesirable immune cells. It is through the association of FAS with the FADD protein that apoptosis is activated.
In the study published in this issue, the team identified heterozygous mutations in the FADD gene in four unrelated patients with ALPS who did not have a mutation in the FAS gene. The FADD gene mutation was also detected in one of the two parents, although he had never shown any symptoms. The researchers demonstrated that both copies of the FADD gene are mutated in the patients' abnormal T lymphocytes. In the four patients described here, the mutation only affects the two copies of FADD in the immune system, since the DUP took place in T lymphocyte progenitors. Other cells (notably heart and neuronal cells) have only one mutated copy of the FADD gene. Mutations in the FADD gene therefore affect the efficiency of apoptosis only in the lymphocytes of these patients. In patients suffering from a much more severe disease affecting the development of several organs (heart, spleen, neurons), mutations inherited from both copies of FADD (and present in all the body's cells) had previously been observed, indicating that apoptosis is also important beyond the immune system.
This innovative study highlights the unusual way in which a genetic disease is expressed across generations. Uniparental disomy, this atypical genetic mechanism, explains why individuals carrying the same mutation may or may not develop the associated disease. There are many genetic diseases with variable expression (no symptoms or very different symptoms from one patient to another). This study shows that somatic mutations, in addition to inherited mutations, should be sought for genetic diseases, particularly autoimmune diseases. Identifying the genetic cause provides a better understanding of the associated disease and, above all, enables targeted and specific treatments to be proposed.